Council Agrees on Mutual Recognition of Covid-19 Test Results Across EU
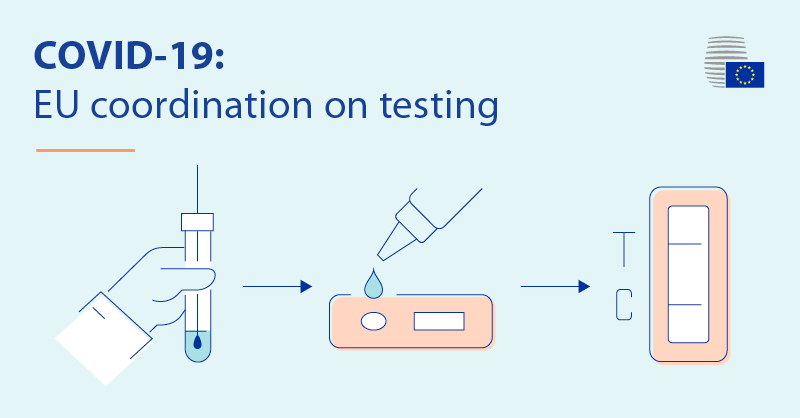
Member states on Thursday unanimously agreed on a Council recommendation setting a common framework for the use of rapid antigen tests and the mutual recognition of coronavirus (Covid-19) test results across the EU.
“This is a central tool to help mitigate the spread of the virus and contribute to the smooth functioning of the internal market,” the Council said in an announcement.
According to the Council, the mutual recognition of test results for the coronavirus infection carried by certified health bodies is essential in order to facilitate cross-border movement, cross-border contact tracing and treatment.
Key components of the recommendation include:
– the validation and mutual recognition of rapid antigen tests and RT-PCR tests among member states
– the sharing of a standardised set of data (through a digital platform)
– the development of a common list of Covid-19 rapid antigen tests
– the prioritisation of situations for the use of such tests (e.g. contacts of confirmed cases, outbreak clusters) and more.
The recommendation also includes future-proof provisions to address the challenges of the evolving pandemic: the common list of appropriate Covid-19 rapid antigen tests should be flexible enough for addition or removal of those tests whose efficacy is impacted by COVID-19 mutations.
The Council Recommendation is not legally binding; it sets out best practices, which member states are encouraged to follow.